Developmental Mechanism - Epithelial Mesenchymal Transition
| Embryology - 23 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The term epithelial mesenchymal transition (EMT) refers to a developmental process where an established epithelium either "breaks down" or "delaminates" allowing cells to leave the epithelium and become connective tissue (mesenchymal) in organisation. This transition can be a permanent change, or a transient event, where the mesenchymal cells may reestablish a new epithelial organisation (mesenchymal epithelial transition).
Epithelial cells (organised cellular layer) which loose their organisation and migrate/proliferate as a mesenchymal cells (disorganised cellular layers) are said to have undergone an Epithelial Mesenchymal Transition (EMT).
Mesenchymal cells, connective tissue-like, that have undergone this process may at a later time and under specific signaling can undergo the opposite process, mesenchyme to epithelia. In development, this process can be repeated several times during tissue differentiation. For example, within the mesoderm somite epithelium, the sclerotome component undergoes EMT forming the skeletal elements of the vertebrae and ribs. Other examples occur in neural crest formation, heart valve formation and Müllerian duct regression.
This process is also studied in carcinogenesis (oncogenesis) or cancer development, where part of this process can be the transformation of an epithelial cell into a mesenchymal cell.[1][2]
Historically, in the late 1970s Elizabeth Hay began studied “epithelial–mesenchymal transformation” in embryogenesis.[3]
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Epithelial Mesenchymal Transition | TWIST | ZEB | SNAIL |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
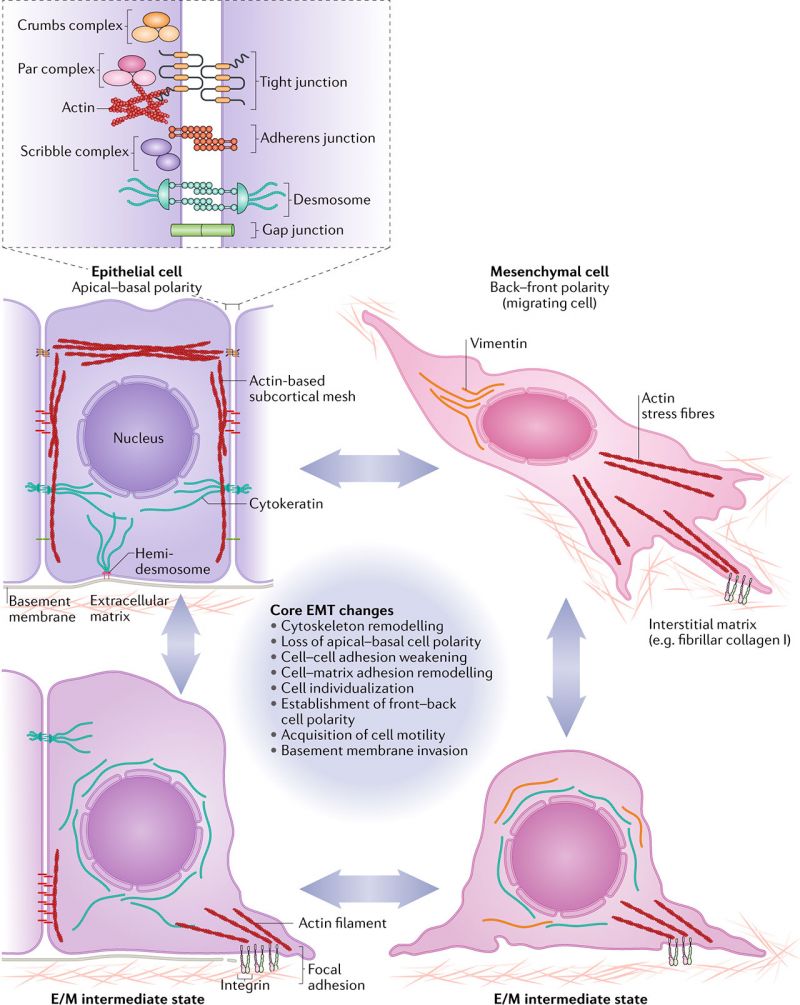
Cell Junction Changes
Epithelial cells are connected by many different cell junctions, that mesenchymamal cells either lack or redistribute. Epithelial cell junctions include adherens junctions, desmosomes, gap junctions and tight junctions.
Mesenchymal cells interact with extracellular matrix and establish polarity in actin stress fibres and intermediate filament focal adhesions.
Epithelial or Mesenchymal Cell State[8]
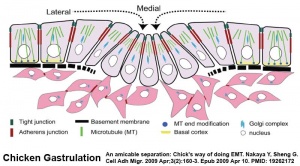
Gastrulation
- Links: gastrulation
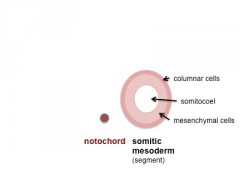
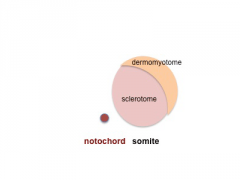
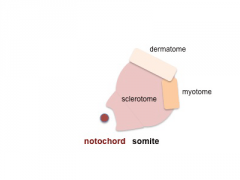
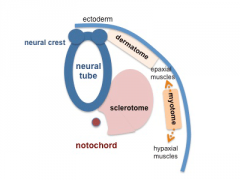
Somite Development
The paraxial mesoderm segments to form somites The sclerotome component of each somite undergoes EMT grating away and eventually forming the axial skeletal elements of the Template:Vertebrae and ribs.
Note - the cartoons show just the embryo righthand side mesoderm development (the same events occur on the lefthand side).
- Somite Links: 1 paraxial | 2 early somite | 3 sclerotome and dermomyotome | 4 dermatome and myotome | 5 somite spreading | SEM image - Human Embryo (week 4) showing somites | Movie - somitogenesis Hes expression
- Somite Cartoons
Cite this page: Hill, M.A. (2024, May 23) Embryology Developmental Mechanism - Epithelial Mesenchymal Transition. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Mechanism_-_Epithelial_Mesenchymal_Transition
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
- Links: somite
Neural Crest Development
Early neural crest cell development migration involves an initial epithelial mesenchymal transition to delaminate from the ectoderm neural tube layer.[9] A microRNA miR-203, acting as part of an epigenetic-microRNA regulatory network, appears influence the timing of this neural crest delamination.[10]
- Links: neural crest
Heart Development
During heart development endocardial and epicardial cells produce non-cardiomyocyte lineages undergo rounds of epithelial to mesenchymal transition, see review.[11]
Palate Development
During the embryonic period the primary palate fusion, between maxillary process and the frontonasal prominence, requires loss of the epithelial seam.

|
- Links: palate
Respiratory Development
| Neonatal Human | Fetal Rabbit |
|---|---|
|
|
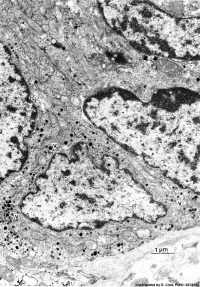
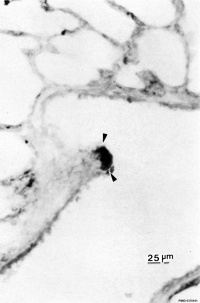
| Pulmonary neuroendocrine cell (EM)[13] | Neuroepithelial body[13] |
Pulmonary Neuroendocrine Cells (PNECs) differentiate in the airway epithelium in late embryonic to early fetal period.[14][15] Later in the mid-fetal period clusters of these cells form neuroepithelial bodies (NEBs). The cells migrate to to form these clusters by a process involving transient epithelial to mesenchymal transition. The process of migration has recently been described as “slithering”[16], where the cells transiently lose epithelial characteristics but remain associated with the membrane while traversing neighboring epithelial cells to reach cluster sites.
- Links: Endocrine Respiratory | respiratory
Molecular
TWIST, ZEB, SNAIL
Twist
The twist family (TWIST1, TWIST2) of basic helix-loop-helix transcription factors are expressed in embryonic mesoderm and have been shown to be key regulators of the epithelial-mesenchymal transition. These transcription factors recognize a consensus DNA element called the E box. Mouse and human TWIST share 96.6% amino acid identity.
ZEB
The Zinc Finger E Box-Binding Homeobox (ZEB) family of 2-handed zinc finger/homeodomain proteins and functions as a DNA-binding transcriptional repressor that interacts with activated SMADs, the transducers of TGF-beta signaling.
Expression of ZEB1 in human epithelial cells can cause a morphologic change from an epithelial to a mesenchymal phenotype.[17]
Mesenchymal-to-Epithelial Transition
The alternate process involves the conversion of the embryonic connective tissue organization (mesenchyme) to an epithelial organization (epithelium) that can occur during developmental processes.
This process can be seen occurring during early somitogenesis.
It is also suggested that this mechanism occurs in the maternal uterus during endometrial regeneration following decidualization.[18][19]
References
- ↑ Savagner P. (2010). The epithelial-mesenchymal transition (EMT) phenomenon. Ann. Oncol. , 21 Suppl 7, vii89-92. PMID: 20943648 DOI.
- ↑ Tseng CH, Murray KD, Jou MF, Hsu SM, Cheng HJ & Huang PH. (2011). Sema3E/plexin-D1 mediated epithelial-to-mesenchymal transition in ovarian endometrioid cancer. PLoS ONE , 6, e19396. PMID: 21559368 DOI.
- ↑ Hay ED. (1995). An overview of epithelio-mesenchymal transformation. Acta Anat (Basel) , 154, 8-20. PMID: 8714286
- ↑ Zhang Z, Wu W, Fang X, Lu M, Wu H, Gao C & Xia Z. (2020). Sox9 promotes renal tubular epithelial‑mesenchymal transition and extracellular matrix aggregation via the PI3K/AKT signaling pathway. Mol Med Rep , , . PMID: 32901875 DOI.
- ↑ Hernández-Martínez R, Ramkumar N & Anderson KV. (2019). p120-catenin regulates WNT signaling and EMT in the mouse embryo. Proc. Natl. Acad. Sci. U.S.A. , 116, 16872-16881. PMID: 31371508 DOI.
- ↑ Martyn I, Kanno TY, Ruzo A, Siggia ED & Brivanlou AH. (2018). Self-organization of a human organizer by combined Wnt and Nodal signalling. Nature , 558, 132-135. PMID: 29795348 DOI.
- ↑ Rinon A, Molchadsky A, Nathan E, Yovel G, Rotter V, Sarig R & Tzahor E. (2011). p53 coordinates cranial neural crest cell growth and epithelial-mesenchymal transition/delamination processes. Development , 138, 1827-38. PMID: 21447558 DOI.
- ↑ Yang J, Antin P, Berx G, Blanpain C, Brabletz T, Bronner M, Campbell K, Cano A, Casanova J, Christofori G, Dedhar S, Derynck R, Ford HL, Fuxe J, García de Herreros A, Goodall GJ, Hadjantonakis AK, Huang RJY, Kalcheim C, Kalluri R, Kang Y, Khew-Goodall Y, Levine H, Liu J, Longmore GD, Mani SA, Massagué J, Mayor R, McClay D, Mostov KE, Newgreen DF, Nieto MA, Puisieux A, Runyan R, Savagner P, Stanger B, Stemmler MP, Takahashi Y, Takeichi M, Theveneau E, Thiery JP, Thompson EW, Weinberg RA, Williams ED, Xing J, Zhou BP & Sheng G. (2020). Guidelines and definitions for research on epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. , , . PMID: 32300252 DOI.
- ↑ Szabó A & Mayor R. (2018). Mechanisms of Neural Crest Migration. Annu. Rev. Genet. , 52, 43-63. PMID: 30476447 DOI.
- ↑ Sánchez-Vásquez E, Bronner ME & Strobl-Mazzulla PH. (2019). Epigenetic inactivation of miR-203 as a key step in neural crest epithelial-to-mesenchymal transition. Development , 146, . PMID: 30910825 DOI.
- ↑ von Gise A & Pu WT. (2012). Endocardial and epicardial epithelial to mesenchymal transitions in heart development and disease. Circ. Res. , 110, 1628-45. PMID: 22679138 DOI.
- ↑ Diewert VM & Lozanoff S. (1993). A morphometric analysis of human embryonic craniofacial growth in the median plane during primary palate formation. J. Craniofac. Genet. Dev. Biol. , 13, 147-61. PMID: 8227288
- ↑ 13.0 13.1 DiAugustine RP & Sonstegard KS. (1984). Neuroendocrinelike (small granule) epithelial cells of the lung. Environ. Health Perspect. , 55, 271-95. PMID: 6376101
- ↑ Cutz E. (1982). Neuroendocrine cells of the lung. An overview of morphologic characteristics and development. Exp. Lung Res. , 3, 185-208. PMID: 6188605
- ↑ Cutz E, Gillan JE & Bryan AC. (1985). Neuroendocrine cells in the developing human lung: morphologic and functional considerations. Pediatr. Pulmonol. , 1, S21-9. PMID: 3906540
- ↑ Kuo CS & Krasnow MA. (2015). Formation of a Neurosensory Organ by Epithelial Cell Slithering. Cell , 163, 394-405. PMID: 26435104 DOI.
- ↑ Vandewalle C, Comijn J, De Craene B, Vermassen P, Bruyneel E, Andersen H, Tulchinsky E, Van Roy F & Berx G. (2005). SIP1/ZEB2 induces EMT by repressing genes of different epithelial cell-cell junctions. Nucleic Acids Res. , 33, 6566-78. PMID: 16314317 DOI.
- ↑ Cousins FL, Murray A, Esnal A, Gibson DA, Critchley HO & Saunders PT. (2014). Evidence from a mouse model that epithelial cell migration and mesenchymal-epithelial transition contribute to rapid restoration of uterine tissue integrity during menstruation. PLoS ONE , 9, e86378. PMID: 24466063 DOI.
- ↑ Patterson AL, Zhang L, Arango NA, Teixeira J & Pru JK. (2013). Mesenchymal-to-epithelial transition contributes to endometrial regeneration following natural and artificial decidualization. Stem Cells Dev. , 22, 964-74. PMID: 23216285 DOI.
Textbooks
Reviews
Wan Y, Liu H, Zhang M, Huang Z, Zhou H, Zhu Y, Tao Y, Xie N, Liu X, Hou J & Wang C. (2020). Prognostic value of epithelial-mesenchymal transition-inducing transcription factors in head and neck squamous cell carcinoma: A meta-analysis. Head Neck , 42, 1067-1076. PMID: 32048783 DOI.
Yang J, Antin P, Berx G, Blanpain C, Brabletz T, Bronner M, Campbell K, Cano A, Casanova J, Christofori G, Dedhar S, Derynck R, Ford HL, Fuxe J, García de Herreros A, Goodall GJ, Hadjantonakis AK, Huang RJY, Kalcheim C, Kalluri R, Kang Y, Khew-Goodall Y, Levine H, Liu J, Longmore GD, Mani SA, Massagué J, Mayor R, McClay D, Mostov KE, Newgreen DF, Nieto MA, Puisieux A, Runyan R, Savagner P, Stanger B, Stemmler MP, Takahashi Y, Takeichi M, Theveneau E, Thiery JP, Thompson EW, Weinberg RA, Williams ED, Xing J, Zhou BP & Sheng G. (2020). Guidelines and definitions for research on epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. , , . PMID: 32300252 DOI.
Thomson TM, Balcells C & Cascante M. (2019). Metabolic Plasticity and Epithelial-Mesenchymal Transition. J Clin Med , 8, . PMID: 31277295 DOI.
Lamouille S, Xu J & Derynck R. (2014). Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. , 15, 178-96. PMID: 24556840 DOI.
Botchkarev VA & Kishimoto J. (2003). Molecular control of epithelial-mesenchymal interactions during hair follicle cycling. J. Investig. Dermatol. Symp. Proc. , 8, 46-55. PMID: 12894994 DOI.
Articles
Zhang M, Wang Y, Matyunina LV, Akbar A & McDonald JF. (2020). The ability of miRNAs to induce mesenchymal-to-epithelial transition (MET) in cancer cells is highly dependent upon genetic background. Cancer Lett. , 480, 15-23. PMID: 32234315 DOI.
Hernández-Martínez R, Ramkumar N & Anderson KV. (2019). p120-catenin regulates WNT signaling and EMT in the mouse embryo. Proc. Natl. Acad. Sci. U.S.A. , 116, 16872-16881. PMID: 31371508 DOI.
Sánchez-Vásquez E, Bronner ME & Strobl-Mazzulla PH. (2019). Epigenetic inactivation of miR-203 as a key step in neural crest epithelial-to-mesenchymal transition. Development , 146, . PMID: 30910825 DOI.
Search PubMed
Search Pubmed: Epithelial Mesenchymal Transition
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- EMT International Association TEMTIA
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 23) Embryology Developmental Mechanism - Epithelial Mesenchymal Transition. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Mechanism_-_Epithelial_Mesenchymal_Transition
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G