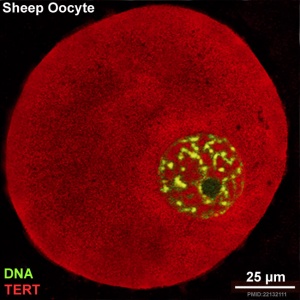
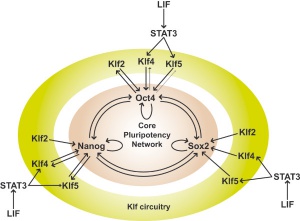
Timeline - Human Embryonic Stem Cell Research
| Embryology - 20 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
A summary timeline of research that led to the discovery and application of human embryonic stem cells. Timeline as shorter print version
1878
First reported attempts to fertilize mammalian eggs outside the body
1959
First report of animals (rabbits) produced through IVF in the United States
1960's
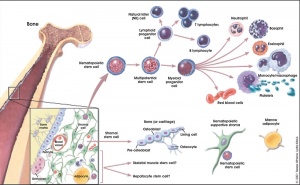
Studies of teratocarcinomas in the testes of several inbred strains of mice indicates they originated from embryonic germ cells. The work establishes embryonal carcinoma (EC) cells as a kind of stem cell
1968
Edwards and Bavister fertilize the first human egg in vitro
1970's
EC cells injected into mouse blastocysts produce chimeric mice. Cultured SC cells are explored as models of embryonic development, although their complement of chromosomes is abnormal
1978
Louise Brown, the first IVF baby, is born in England
1980
Australia's first IVF baby, Candace Reed, is born in Melbourne
1981
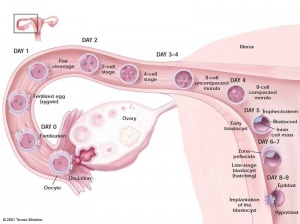
Evans and Kaufman, and Martin derive mouse embryonic stem (ES) cells from the inner cell mass of blastocysts. They establish culture conditions for growing pluripotent mouse ES cells in vitro. The ES cells yield cell lines with normal, diploid karyotyes and generate derivatives of all three primary germ layers as well as primordial germ cells. Injecting the ES cells into mice induces the formation of teratomas. The first IVF baby, Elizabeth Carr, is born in the United States.
1984–88
Andrews et al., develop pluripotent, genetically identical (clonal) cells called embryonal carcinoma (EC) cells from Tera-2, a cell line of human testicular teratocarcinoma. Cloned human teratoma cells exposed to retinoic acid differentiate into neuron-like cells and other cell types
1989
Pera et al., derive a clonal line of human embryonal carcinoma cells, which yields tissues from all three primary germ layers. The cells are aneuploid (fewer or greater than the normal number of chromosomes in the cell) and their potential to differentiate spontaneously in vitro is typically limited. The behavior of human EC cell clones differs from that of mouse ES or EC cells
1994
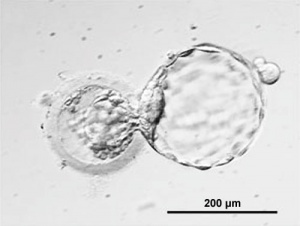
Human blastocysts created for reproductive purposes using IVF and donated by patients for research, are generated from the 2-pronuclear stage. The inner cell mass of the blastocyst is maintained in culture and generates aggregates with trophoblast-like cells at the periphery and ES-like cells in the center. The cells retain a complete set of chromosomes (normal karyotype); most cultures retain a stem cell-like morphology, although some inner cell mass clumps differentiate into fibroblasts. The cultures are maintained for two passages
1995–96
Non-human primate ES cells are derived and maintained in vitro, first from the inner cell mass of rhesus monkeys, and then from marmosets. The primate ES cells are diploid and have normal karyotypes. They are pluripotent and differentiate into cells types derived from all three primary germ layers. The primate ES cells resemble human EC cells and indicate that it should be possible to derive and maintain human ES cells in vitro.
1998
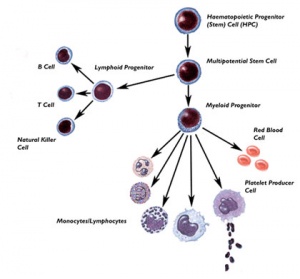
Thomson et al., derive human ES cells from the inner cell mass of normal human blastocysts donated by couples undergoing treatment for infertility. The cells are cultured through many passages, retain their normal karyotypes, maintain high levels of telomerase activity, and express a panel of markers typical of human EC cells non-human primate ES cells. Several (non-clonal) cell lines are established that form teratomas when injected into immune-deficient mice. The teratomas include cell types derived from all three primary germ layers, demonstrating the pluripotency of human ES cells. Gearhart and colleagues derive human embryonic germ (EG) cells from the gonadal ridge and mesenchyma of 5- to 9-week fetal tissue that resulted from elective abortions. They grow EG cells in vitro for approximately 20 passages, and the cells maintain normal karyotypes. The cells spontaneously form aggregates that differentiate spontaneously, and ultimately contain derivatives of all three primary germ layers. Other indications of their pluripotency include the expression of a panel of markers typical of mouse ES and EG cells. The EG cells do not form teratomas when injected into immune-deficient mice
2000
Scientists in Singapore and Australia led by Pera, Trounson, and Bongso derive human ES cells from the inner cell mass of blastocysts donated by couples undergoing treatment for infertility. The ES cells proliferate for extended periods in vitro, maintain normal karyotypes, differentiate spontaneously into somatic cell lineages derived from all three primary germ layers, and form teratomas when injected into immune-deficient mice.
2001
As human ES cell lines are shared and new lines are derived, more research groups report methods to direct the differentiation of the cells in vitro. Many of the methods are aimed at generating human tissues for transplantation purposes, including pancreatic islet cells, neurons that release dopamine, and cardiac muscle cells.
2011
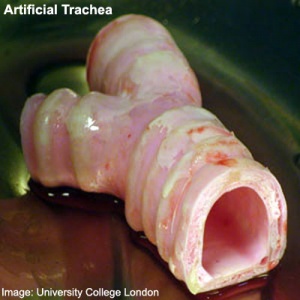
First Successful Transplantation of a Synthetic Tissue Engineered Trachea (Windpipe)
- An international team designed and built the nanocomposite tracheal scaffold and produced a specifically designed bioreactor used to seed the scaffold with the patient´s own stem cells. The cells were grown on the scaffold inside the bioreactor for two days before transplantation to the patient. Because the cells used to regenerate the trachea were the patient's own, there has been no rejection of the transplant and the patient is not taking immunosuppressive drugs.
- Karolinska Institute | University College London | BBC News
2012
- The Nobel Prize in Physiology or Medicine 2012 was awarded jointly to Sir John B. Gurdon and Shinya Yamanaka "for the discovery that mature cells can be reprogrammed to become pluripotent" (More? Yamanaka Factors | Induced Stem Cells | Stem Cells | Nobel Prize)
- Yamanaka Factors Are a set of 4 transcription factors when introduced into cells induces stem cell formation. PMID 16904174 | PMID 18035408 | PMID 20535199
- John Gurdon used nuclear transplantation and cloning to show that the nucleus of a differentiated somatic cell retains the totipotency necessary to form a whole organism. 2003 Current Biology Interview PMID 14521852 2009 Interview - "The birth of cloning" PMID 19132124
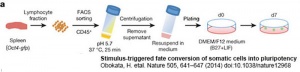
2014
- Stimulus-triggered fate conversion of somatic cells into pluripotency disproved - RIKEN Panel Finds Misconduct in Reprogrammed Stem Cell Papers Science April 2014 |
- Japanese research institute has opened an investigation into this groundbreaking stem cell study after concerns were raised about its credibility. The RIKEN investigation follows allegations on blog sites about the use of duplicated images in Obokata’s papers, and numerous failed attempts to replicate her results. Nature
2015
Some timeline information modified from NIH - Stem Cells: Scientific Progress and Future Research Directions 2001
References
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 20) Embryology Timeline - Human Embryonic Stem Cell Research. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Timeline_-_Human_Embryonic_Stem_Cell_Research
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G