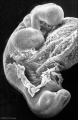
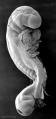
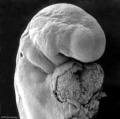
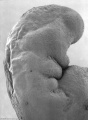
Abnormal Development - Thalidomide
| Embryology - 19 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
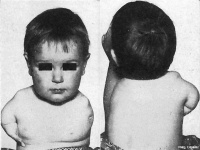
The drug thalidomide (Contergan, Distaval) was introduced on to the market on October 1, 1957 in West Germany. Thalidomide soon became a drug prescribed to pregnant women to combat symptoms associated with morning sickness. In West Germany, thalidomide could be bought without a prescription. In the United Kingdom, thalidomide was first marketed in April, 1958 (and later withdrawn from sale in December, 1961).[2] When taken during the first trimester of pregnancy, thalidomide prevented the proper growth of the fetus resulting in horrific birth defects ("thalidomide embryopathy") in thousands of children around the world. When taken, mainly in first world countries, between day 20 to 36 after fertilisation (GA 34–50 days LMP) children were born with limb and other defects. In the late 1950's and early 1960's these children became known as "Thalidomide babies".

|

|
| Dr Widukind Lenz | Dr William McBride |
| In Germany, a 1961 report by Dr Widukind Lenz described abnormalities with "Contergan".[3]
(More? Historic People) |
In Australia, a brief letter by Dr William McBride, linked "Distaval" to newborn abnormalities.[4] |
Not all species embryos are affected by the drug in the same way, with human and rabbit being most susceptible to the teratogenic effects. In addition, the effect on human development is also dependent upon the time and dose of the drug exposure, the "critical periods".
More recently there has been a clinical revival for thalidomide use in non-pregnant women, during cancer chemotherapy[5][6], treatment for multiple myeloma and graft rejection.[7].
- Thalidomide Links: limb | limb abnormalities | hearing abnormalities | vision abnormalities | musculoskeletal abnormalities | K12 Thalidomide
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Thalidomide Teratology | Thalidomide | Cereblon | Tbx5 |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Nausea and Vomiting of Pregnancy
Around half to two-thirds of all pregnant women will experience nausea and vomiting of pregnancy, typically called "morning sickness". Occurring mainly in the first trimester due to several possible identified causes that include; changed and high levels of hormones, blood pressure fluctuations and changes in carbohydrate metabolism. The more severe prolonged vomiting clinical form is described as hyperemesis gravidarum.
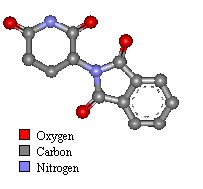
Thalidomide Physical and Chemical Properties
Thalidomide was first synthesised in 1954 by Wilhelm Kunz, a German drug discovery pharmacist for Chemie Grünenthal, searching for new organic compounds. The drug was manufactured as a two enantiomer isomer mix (laevo+ and dextro-). The teratogenic form was laevo+ thalidomide, though rabbits appear to be sensitive to both forms.
- Molecular Mass: 258.23 Da
- Color: white crystalline
- Odor: Odorless
- Taste: taste-less
- Melting point: 271°C.
- Insoluble in ether and benzene
- Has a low solubility in water, methanol, ethanol and glacial acetic acid
| Historic 1960 Pharmacology |
|---|
Pharmacological properties of thalidomide (alpha-phthalimido glutarimide), a new sedative hypnotic drug (1960)[16]
Study was carried out on mice, rats and anaesthetised cats. |
Historic People
Germany

|
Dr Widukind Lenz (1919 - 1995) was a German paediatrician who in 1961 identified the association of thalidomide ("Contergan") with birth abnormalities.[17][3][18]
Dr Lenz was physician-in-chief of the Eppendorfer Kinderklinik (1952), then chair of paediatrics at the University of Hamburg (1961), and director of the Institute of Human Genetics in Münster (1965). |
| Dr Widukind Lenz |
In Germany it is estimated that between 1957 and 1962, approximately 5000 children were born with severe birth defects due to maternal thalidomide use as a sedative and as effective relief from morning sickness.[19] A recent study by researchers of this German population, has attempted to generate reference tables of limb to body height ratios of healthy human adults for assessing potential thalidomide embryopathy.[20]
Australia

|
A brief letter by an Australian clinician, William McBride, identified "Distaval" as a teratogenic agent, linking of newborn abnormalities with the taking of thalidomide causing a "thalidomide embryopathy"[4]. Later in 1981, Dr McBride attempted to show similar teratogenic effects of a second "Debendox", resulting in a scientific case related to his falsification of data. The second drug has since been shown to not be associated with teratogenic effects.[21] |
| Dr William McBride |
USA
Helen Brooke Taussig
Dr Helen Brooke Taussig (1898-1986) was a paediatric cardiologist, who developed a surgical procedure for Tetralogy of Fallot, also campaigned for blocking introduction of thalidomide into the U.S.A.[22] One of her students had drawn her attention to the data on congenital malformations occurring in Germany and England.
- Links: NLM
Frances Oldham Kelsey
Frances Oldham Kelsey (1914 - 2015) was at the Food and Drug Administration, where she reviewed of a sleeping pill based on thalidomide already widely used in Europe, but Kelsey was concerned by some data suggesting dangerous side effects in patients who took the drug repeatedly. She continued to withhold drug approval for the U.S.A. leading to less cases in that country.
NLS Changing the Shape of Medicine
- "In 1960 she moved to Washington, D.C., and began her long and distinguished career at the Food and Drug Administration, where she later became chief of the Division of New Drugs, director of the Division of Scientific Investigations, and deputy for Scientific and Medical Affairs, Office of Compliance. Dr. Frances Kelsey took her stand against thalidomide during her first month at the Food and Drug Administration, on her first assignment. The task was supposed to be a straightforward review of a sleeping pill already widely used in Europe, but Kelsey was concerned by some data suggesting dangerous side effects in patients who took the drug repeatedly. While she continued to withhold approval, the manufacturers tried everything they could to get around her judgement."
Frances Oldham Kelsey (1914 - 2015) Food and Drug Administration officer who saved U.S. babies from thalidomide dies at 101.
- Links: The New York Times NIH Biography
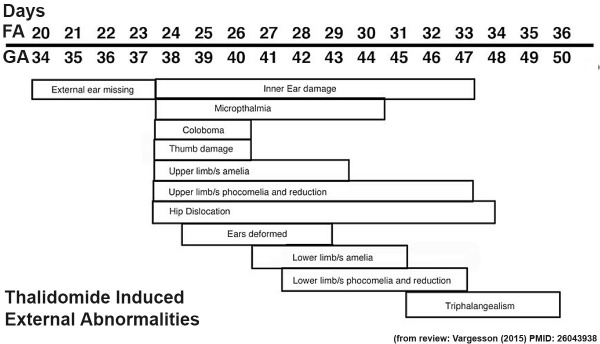
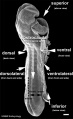
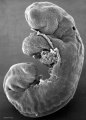
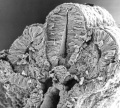
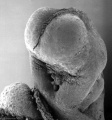
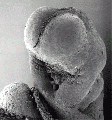
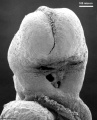
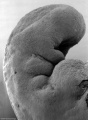
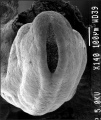
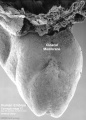
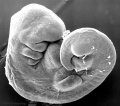
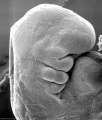
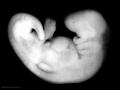
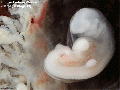
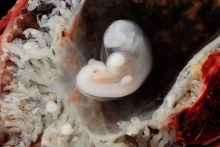
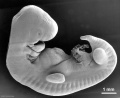
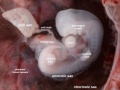
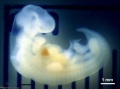
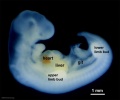
Thalidomide External Effects
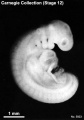
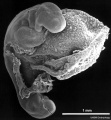
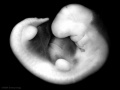
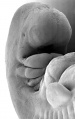
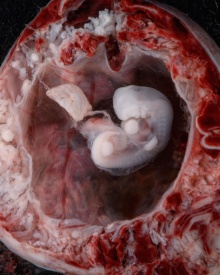
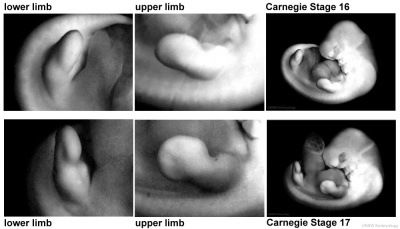
Period approximately covers the developmental stages 11 to 15.
| Week: | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Carnegie stage: | 1 2 3 4 | 5 6 | 7 8 9 | 10 11 12 13 | 14 15 | 16 17 | 18 19 | 20 21 22 23 |
Thalidomide external effects timeline[23]
|
|
|
|
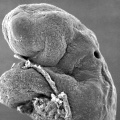
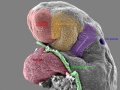
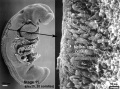
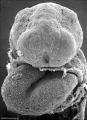
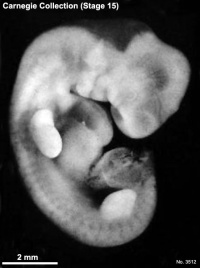
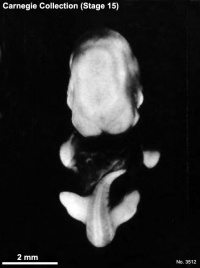
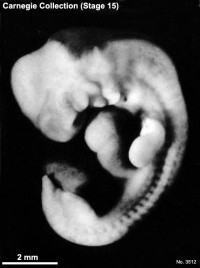
Carnegie stage 15
35 - 38 days
Limb development after the sensitive period
|
|
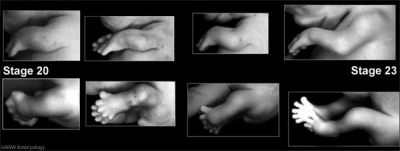
| Human embryonic limb development (week 6) | Human embryonic limb development (week 8) |
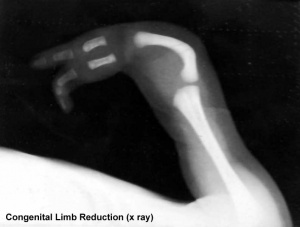
Limb Reduction
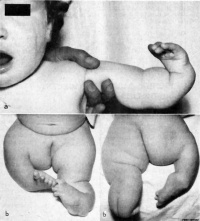
Text extract below from [24], note that all timings are using the clinical dates of Last Menstrual Period (LMP, GA), which differ by about 14 days more to the developmental date from fertilization.
"It has been suggested that thalidomide does not produce malformations if only taken before the 34th day after the last menstruation (LMP) and usually no malformation if taken only after the 50th day.
Within the sensitive period from day 35 to day 49, there is the following sequence:
- 35th - 37th day Absence of the ears and deafness
- 39th - 41st day Absence of arms
- 43rd - 44th day Phocomelia with three fingers
- 46th - 48th day Thumbs with three joints
If thalidomide was taken throughout the sensitive period, the consequence may be severe defects of ears, arms and legs and of internal malformations, which often led to early death. About 40% of thalidomide victims died before their first birthday."
These are W. Lenz's historic observations and relate to easily identifiable features.
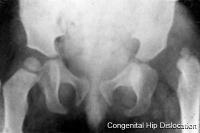
Hip Dislocation
Microphthalmia
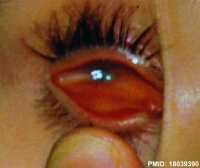
Microphthalmia clinical image[25] |
A clinical description for the presence of a small eye located within the orbit. This condition occurs in up to 11% of blind children. |
Hearing Abnormalities
Hearing loss associated with abnormalities of the inner ear development (otic placode origin) and external ear reduction or deformation (pharyngeal arch origin).
- Otic Placode Development
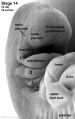
| Pharyngeal Arch Development (external ear, stages 14-23 and adult) | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
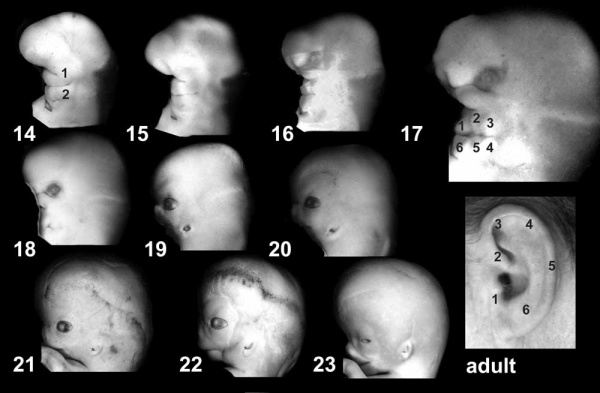
|
|
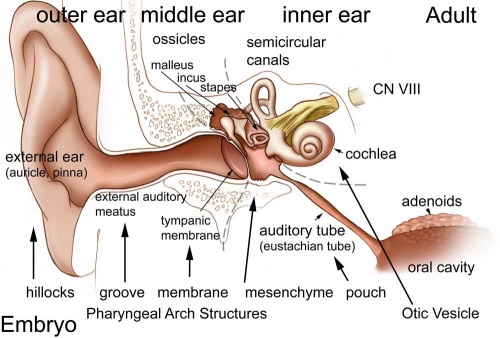
Adult hearing embryonic origins |
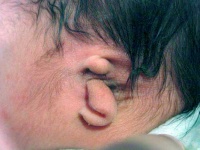
External Ear - Microtia |
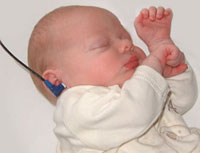
Newborn hearing test |
Cereblon
Cereblon (CRBN) was named based on its possible role in cerebral development and the presence of a Lon protease domain. Functionally it had been previously identified as being associated with neural development. Thalidomide specifically binds cereblon, which inhibits the ubiquitin ligase activity of the SCF protein ligase complex, possibly leading to abnormal regulation of the BMP and FGF8 signaling pathways. The SCF complex (Skp, Cullin, F-box containing complex) is a multi-protein E3 ubiquitin ligase complex catalyzing the ubiquitination of proteins destined for proteasomal degradation.
- thalidomide binds to CRBN at specific sites.
- interaction disrupts the function of the E3 ubiquitin ligase complex (composed by proteins CRBN, DDB1, and Cul4)
- down-regulation of fibroblast growth factor genes.
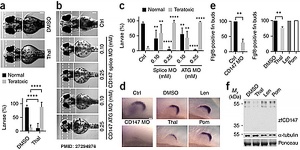
Immunomodulatory drugs disrupt the cereblon-CD147-MCT1 axis to exert antitumor activity and teratogenicity[8] "Immunomodulatory drugs (IMiDs), such as thalidomide and its derivatives lenalidomide and pomalidomide, are key treatment modalities for hematologic malignancies, particularly multiple myeloma (MM) and del(5q) myelodysplastic syndrome (MDS). Cereblon (CRBN), a substrate receptor of the CRL4 ubiquitin ligase complex, is the primary target by which IMiDs mediate anticancer and teratogenic effects. Here we identify a ubiquitin-independent physiological chaperone-like function of CRBN that promotes maturation of basigin (BSG; also known as CD147) and solute carrier family 16 member 1 (SLC16A1; also known as MCT1) proteins. This process allows for the formation and activation of the CD147-MCT1 transmembrane complex, which promotes various biological functions, including angiogenesis, proliferation, invasion and lactate export. We found that IMiDs outcompete CRBN for binding to CD147 and MCT1, leading to destabilization of the CD147-MCT1 complex. Accordingly, IMiD-sensitive MM cells lose CD147 and MCT1 expression after being exposed to IMiDs, whereas IMiD-resistant cells retain their expression. Furthermore, del(5q) MDS cells have elevated CD147 expression, which is attenuated after IMiD treatment. Finally, we show that BSG (CD147) knockdown phenocopies the teratogenic effects of thalidomide exposure in zebrafish. These findings provide a common mechanistic framework to explain both the teratogenic and pleiotropic antitumor effects of IMiDs." [[Abnormal Development - Thalidomide#CD147 (Basigin)|CD147 (Basigin)]
- Links: OMIM - Cereblon | UniProt - CRBN
Mouse Neural Cereblon Expression
The following table is from a study of the levels of cereblon messenger RNA (mRNA) in different regions of the adult mouse brain.[26]
Arbitrary signal strength detected by in situ hybridisation from low (+) to high (+++++).
| Region | Intensity |
|---|---|
| Infralimbic cortex | ++++ |
| Cingulate cortex | ++++ |
| Other neocortex; layer V | ++++ |
| Other neocortex; other layers | +++ |
| Piriform cortex | ++++ |
| Accumbens nucleus | ++ |
| Caudate putamen | ++ |
| Lateral globus pallidus | + |
| Pyramidal layer of the hippocampus | +++++ |
| Granular layer of dentate gyrus | +++++ |
| Habenular nucleus | ++++ |
| Paraventricular hypothalamic nucleus | +++ |
| Thalamus | ++ ∼ +++ |
| Substantia nigra compacta | +++ |
| Red nucleus, magnocellular part | ++++ |
| Raphe nuclei | +++ ∼ ++++ |
| Trigeminal nucleus | +++++ |
| Reticulotegmental nucleus of the pons | ++++ |
| Parvicellular reticular nucleus | +++ |
| Gigantocellular reticular nucleus | ++++ |
| Purkinje layer of the cerebellum | +++++ |
| Granular layer of the cerebellum | ++++ |
Signaling Pathway
Two possible mechanisms:[13]
- cereblon dependent process
- cereblon independent process (producing reactive oxygen species)[27]
Thalidomide shown to:
- inhibits production of some cytokines (tumor necrosis factor–alpha and vascular endothelial growth factor)
- inducing apoptosis and producing reactive oxygen species
- fgf8 is a downstream target
CD147 (Basigin)

(basigin, BSG, CD147, EMMPRIN) Basigin is a member of the immunoglobulin (Ig) superfamily, with a structure related to the putative primordial form of the family. CD147 has recently been shown[8] to be one of the downstream targets of the cereblon singling pathway and down-regulation can cause similar teratogenic effects in a Zebrafish model.
See also a review by Basigin[28] "Basigin has isoforms; the common form (basigin or basigin-2) has two immunoglobulin domains, and the extended form (basigin-1) has three. Basigin is the receptor for cyclophilins, S100A9 and platelet glycoprotein VI, whereas basigin-1 serves as the receptor for the rod-derived cone viability factor. Basigin tightly associates with monocarboxylate transporters and is essential for their cell surface translocation and activities. In the same membrane plane, basigin also associates with other proteins including GLUT1, CD44 and CD98."
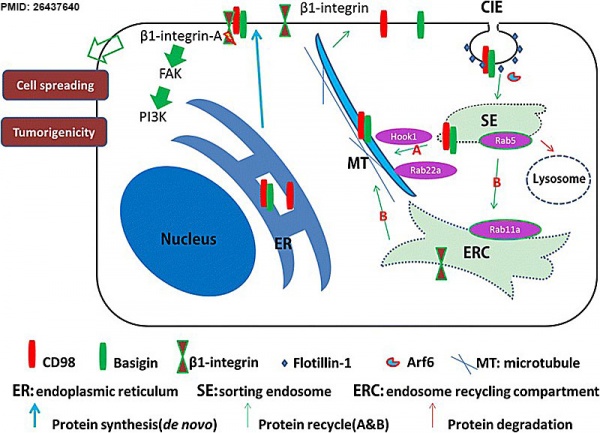
Model for tumorigenic effect of CD147 action on CD98[29]
General model for basigin promotes the membrane localization of CD98 and activation of β1-integrin. Basigin could facilitate the membrane localization of newly synthesized CD98 in the ER (blue arrow). Prototypical CIE cargo proteins enter cells through Arf6-positive endocytic vesicles that either fuse with or mature into Rab5a-positive early endosomes (sorting endosomes), where internalized proteins converge and are sorted for recycling (green arrow) or degradation (red arrow). Basigin and CD98 are internalized by a pathway associated with flotillin-1 and the small G protein Arf6. Thereafter, basigin and CD98 could recycle back to the membrane through the fast recycling pathway (green arrow A) mediated by hook1, Rab22a, microtubules and their own cytoplasmic sequences. Other internalized proteins, including integrins, could recycle through the slow recycling pathway (green arrow B). By promoting the membrane redistribution or translocation of CD98, basigin activates β1 integrins and plays a critical role in liver cancer progression.
- Links: OMIM - Basigin
Vascular Effect
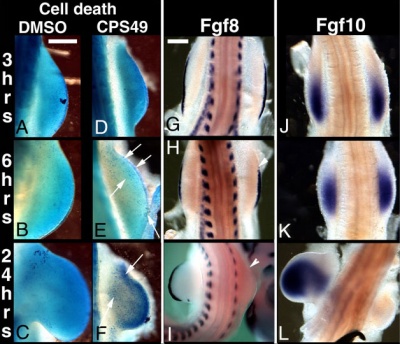
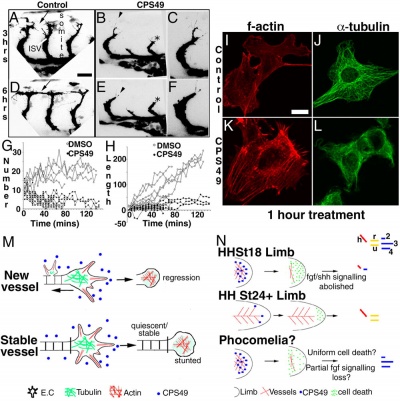
This recent study[15] has used a chicken limb model system and treatment with a chemical, CPS49 a tetrafluorinated analogue of thalidomide that is chemically and structurally related to thalidomide breakdown products.
|
|
| Chicken limb development signaling[15] | Thalidomide - CPS49 vascular effect on chicken model[15] |
The researchers identified an antiangiogenic activity, inhibition of blood vessel growth, of CPS49 within the developing limb and in a number of in vitro tissue culture models of vascular growth. The vessel loss was described as the "primary trigger" leading to an increased cell death and impairment in limb signaling pathways, resulting consequently in both limb outgrowth failure and limb truncations. The possibility that fibroblast growth factor levels are altered, may also fit in with the specific thalidomide binding to cereblon.
Primate Model
Crab-eating Macaque (Macaca fascicularis, Cynomolgus Monkey, Philippine Monkey, Long-tailed Macaque)[14] "Cynomolgus monkeys were orally administered thalidomide at 15 or 20mg/kg-d on days 26-28 of gestation, and fetuses were examined on day 100-102 of gestation. Limb defects such as micromelia/amelia, paw/foot hyperflexion, polydactyly, syndactyly, and brachydactyly were observed in seven of eight fetuses."
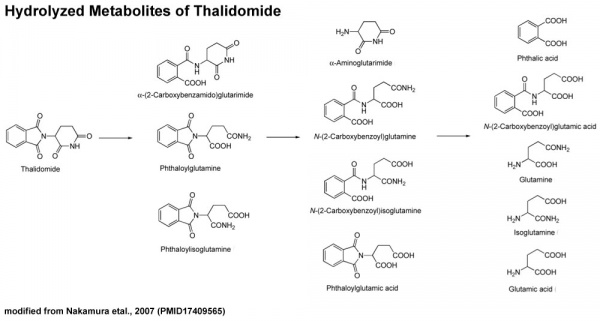
Thalidomide Metabolism in Different Species

It also appears that not all species metabolise thalidomide the same, shown in a study comparing the metabolic breakdown products in mice and man:[31]
- "Our results show that thalidomide metabolite profiles in multiple myeloma patients differ considerably from those in mice. The lack of measurable hydroxylated metabolites in urine and in 1 case plasma of these patients suggests that such metabolites are not responsible for the therapeutic effects of thalidomide in multiple myeloma."
Lenalidomide
(CC-5013, Revlimid) A derivative of thalidomide introduced in 2004, initially intended as a treatment for multiple myeloma. Has also shown efficacy in the hematological disorders, myelodysplastic syndromes. Each year in Australia around 1,500 people are diagnosed with myeloma. Thalidomide can be used in the initial treatment of myeloma and also to control myeloma that has come back (relapsed).
Molecular Formula: C13H13N3O3 Molecular Weight: 259
Advisory Committee on Medicines
- 1963 - Australian Drug Evaluation Committee (ADEC) was established in 1963 following the thalidomide experience.
- 2010 - the ADEC committee was replaced by the Advisory Committee on Prescription Medicines (ACPM)
- 2017 - ACPM committee was replaced by the Advisory Committee on Medicines (ACM).
- The ACM provides independent medical and scientific advice to the Minister for Health and the Therapeutic Goods Administration (TGA) on issues relating to the safety, quality and efficacy of medicines supplied in Australia including issues relating to pre-market and post-market functions for medicines.
| Australian Drug Categories |
|---|
| Legal drugs are classified, usually by each country's appropriate regulatory body, on the safety of drugs during pregnancy. In Australia, the Therapeutic Goods Authority has classes (A, B1, B2, B3, C, D and X) to define their safety. In the USA, drugs are classified by the Food and Drug Administration (FDA) into classes (A, B, C, D, and X) to define their safety. (More? Australian Drug Categories)
|
| Drug Testing |
|---|
| Typical testing of new drug compound today involves a lengthy series of animal and human studies.
Animal studies Usually tested in at least two mammalian species (rats and guinea pigs) using both single and repeated doses. For determining reproductive effects, tests on both male and female animals with dosing begins 4 weeks prior to mating are conducted to determine effects on fertility in both sexes, on embryogenesis, and on fetal malformation. Human Clinical trials Following animal studies to determine dose, efficacy and apparent safety, human studies can commence. Clinical trials are carried out under very strict conditions, set by international regulatory bodies in agreement with the principles espoused in the Declaration of Helsinki. There are four phases to the trials.
|
| After phase I to III the pharmaceutical company compiles all study data for independent assessment by government regulatory authorities in each country.
Regulatory Authorities: FDA in the USA, Therapeutic Goods Administration (TGA) in Australia, Medsafe in New Zealand, Medicines & Healthcare products Regulatory Agency (MHRA) in the UK, and Health Products and Food Branch (HPFB) in Canada. |
| Declaration of Helsinki |
| The Declaration of Helsinki was developed by The World Medical Association (WMA) as a statement of ethical principles for medical research involving human subjects, including research on identifiable human material and data. The Declaration is intended to be read as a whole and each of its constituent paragraphs should not be applied without consideration of all other relevant paragraphs. It is widely regarded as the cornerstone document on human research ethics. It is named after the location of its initial adoption in Helsinki, Finland, in June 1964. |
Teratology
Now consider how different environmental effects during pregnancy may influence developmental outcomes. The terms listed below are often used to describe these environmental effects
- Teratogen (Greek, teraton = monster) any agent that causes a structural abnormality (congenital abnormalities) following fetal exposure during pregnancy. The overall effect depends on dosage and time of exposure. (More? Critical Periods of Development)
- Absolute risk the rate of occurrence of an abnormal phenotype among individuals exposed to the agent. (e.g. fetal alcohol syndrome)
- Relative risk the ratio of the rate of the condition among the exposed and the nonexposed. (e.g. smokers risk of having a low birth weight baby compared to non-smokers) A high relative risk may indicate a low absolute risk if the condition is rare.
- Mutagen a chemical or agent that can cause permanent damage to the deoxyribonucleic acid (DNA) in a cell. DNA damage in the human egg or sperm may lead to reduced fertility, spontaneous abortion (miscarriage), birth defects and heritable diseases.
- Fetotoxicant is a chemical that adversely affects the developing fetus, resulting in low birth weight, symptoms of poisoning at birth or stillbirth (fetus dies before it is born).
- Synergism when the combined effect of exposure to more than one chemical at one time, or to a chemical in combination with other hazards (heat, radiation, infection) results in effects of such exposure to be greater than the sum of the individual effects of each hazard by itself.
- Toxicogenomics the interaction between the genome, chemicals in the environment, and disease. Cells exposed to a stress, drug or toxicant respond by altering the pattern of expression of genes within their chromosomes. Based on new genetic and microarray technologies.
References
- ↑ 1.0 1.1 GILLIS L. (1962). Thalidomide babies: management of limb defects. Br Med J , 2, 647-51. PMID: 13898662
- ↑ LECK IM & MILLAR EL. (1962). Incidence of malformations since the introduction of thalidomide. Br Med J , 2, 16-20. PMID: 14463369
- ↑ 3.0 3.1 LENZ W & KNAPP K. (1962). Thalidomide embryopathy. Arch. Environ. Health , 5, 100-5. PMID: 14464040
- ↑ 4.0 4.1 McBride WG. (1977). Thalidomide embryopathy. Teratology , 16, 79-82. PMID: 331548 DOI.
- ↑ Li PK, Pandit B, Sackett DL, Hu Z, Zink J, Zhi J, Freeman D, Robey RW, Werbovetz K, Lewis A & Li C. (2006). A thalidomide analogue with in vitro antiproliferative, antimitotic, and microtubule-stabilizing activities. Mol. Cancer Ther. , 5, 450-6. PMID: 16505120 DOI.
- ↑ Chuah B, Lim R, Boyer M, Ong AB, Wong SW, Kong HL, Millward M, Clarke S & Goh BC. (2007). Multi-centre phase II trial of Thalidomide in the treatment of unresectable hepatocellular carcinoma. Acta Oncol , 46, 234-8. PMID: 17453375 DOI.
- ↑ Zhang Y, Yang Y, Li X, Chen D, Tang G & Men T. (2019). Thalidomide ameliorate graft chronic rejection in an allogenic kidney transplant model. Int. Immunopharmacol. , 71, 32-39. PMID: 30877871 DOI.
- ↑ 8.0 8.1 8.2 8.3 Eichner R, Heider M, Fernández-Sáiz V, van Bebber F, Garz AK, Lemeer S, Rudelius M, Targosz BS, Jacobs L, Knorn AM, Slawska J, Platzbecker U, Germing U, Langer C, Knop S, Einsele H, Peschel C, Haass C, Keller U, Schmid B, Götze KS, Kuster B & Bassermann F. (2016). Immunomodulatory drugs disrupt the cereblon-CD147-MCT1 axis to exert antitumor activity and teratogenicity. Nat. Med. , 22, 735-43. PMID: 27294876 DOI.
- ↑ Nemer G & Khalil A. (2019). A Cautious Note on Thalidomide Usage in Cancer Treatment: Genetic Profiling of the TBX2 Sub-Family Gene Expression is Required. Drug Res (Stuttg) , , . PMID: 30934098 DOI.
- ↑ Mikami I, Takahashi Y, Koiwa J, Okamura M & Tanaka T. (2019). Zebrafish Yolk Sac Microinjection of Thalidomide for Assessment of Developmental Toxicology. Congenit Anom (Kyoto) , , . PMID: 30932217 DOI.
- ↑ Sorensen D, Sackett A, Urban DJ, Maier J, Vargesson N & Sears KE. (2017). A new mammalian model system for thalidomide teratogenesis: Monodelphis domestica. Reprod. Toxicol. , 70, 126-132. PMID: 28130151 DOI.
- ↑ Ito T & Handa H. (2012). Deciphering the mystery of thalidomide teratogenicity. Congenit Anom (Kyoto) , 52, 1-7. PMID: 22348778 DOI.
- ↑ 13.0 13.1 Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y, Yamaguchi Y & Handa H. (2010). Identification of a primary target of thalidomide teratogenicity. Science , 327, 1345-50. PMID: 20223979 DOI.
- ↑ 14.0 14.1 Ema M, Ise R, Kato H, Oneda S, Hirose A, Hirata-Koizumi M, Singh AV, Knudsen TB & Ihara T. (2010). Fetal malformations and early embryonic gene expression response in cynomolgus monkeys maternally exposed to thalidomide. Reprod. Toxicol. , 29, 49-56. PMID: 19751816 DOI.
- ↑ 15.0 15.1 15.2 15.3 Therapontos C, Erskine L, Gardner ER, Figg WD & Vargesson N. (2009). Thalidomide induces limb defects by preventing angiogenic outgrowth during early limb formation. Proc. Natl. Acad. Sci. U.S.A. , 106, 8573-8. PMID: 19433787 DOI.
- ↑ SOMERS GF. (1960). Pharmacological properties of thalidomide (alpha-phthalimido glutarimide), a new sedative hypnotic drug. Br J Pharmacol Chemother , 15, 111-6. PMID: 13832739
- ↑ LENZ W & KNAPP K. (1962). [Thalidomide embryopathy]. Dtsch. Med. Wochenschr. , 87, 1232-42. PMID: 14464041 DOI.
- ↑ Lenz W. (1985). Thalidomide embryopathy in Germany, 1959-1961. Prog. Clin. Biol. Res. , 163C, 77-83. PMID: 3991661
- ↑ Samel C, Albus C, Tebest R, Forster K, Lüngen M, Niecke A, Pfaff H & Peters KM. (2019). [Upcoming challenges in providing care for thalidomide impaired individuals]. Pflege , , 1-9. PMID: 30887894 DOI.
- ↑ Reimann A, Beyer R, Mumm R & Scheffler C. (2019). Reference tables with centiles of limb to body height ratios of healthy human adults for assessing potential thalidomide embryopathy. Anthropol Anz , , . PMID: 30869735 DOI.
- ↑ Fleming DM, Knox JD & Crombie DL. (1981). Debendox in early pregnancy and fetal malformation. Br Med J (Clin Res Ed) , 283, 99-101. PMID: 6789952
- ↑ TAUSSIG HB. (1962). The thalidomide syndrome. Sci. Am. , 207, 29-35. PMID: 13919872
- ↑ Vargesson N. (2015). Thalidomide-induced teratogenesis: history and mechanisms. Birth Defects Res. C Embryo Today , 105, 140-56. PMID: 26043938 DOI.
- ↑ W. Lenz The History of Thalidomide a lecture given at the 1992 UNITH Congress.
- ↑ Verma AS & Fitzpatrick DR. (2007). Anophthalmia and microphthalmia. Orphanet J Rare Dis , 2, 47. PMID: 18039390 DOI.
- ↑ Aizawa M, Abe Y, Ito T, Handa H & Nawa H. (2011). mRNA distribution of the thalidomide binding protein cereblon in adult mouse brain. Neurosci. Res. , 69, 343-7. PMID: 21241746 DOI.
- ↑ Knobloch J & Rüther U. (2008). Shedding light on an old mystery: thalidomide suppresses survival pathways to induce limb defects. Cell Cycle , 7, 1121-7. PMID: 18418038 DOI.
- ↑ Muramatsu T. (2016). Basigin (CD147), a multifunctional transmembrane glycoprotein with various binding partners. J. Biochem. , 159, 481-90. PMID: 26684586 DOI.
- ↑ Wu B, Wang Y, Yang XM, Xu BQ, Feng F, Wang B, Liang Q, Li Y, Zhou Y, Jiang JL & Chen ZN. (2015). Basigin-mediated redistribution of CD98 promotes cell spreading and tumorigenicity in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. , 34, 110. PMID: 26437640 DOI.
- ↑ Nakamura T, Noguchi T, Miyachi H & Hashimoto Y. (2007). Hydrolyzed metabolites of thalidomide: synthesis and TNF-alpha production-inhibitory activity. Chem. Pharm. Bull. , 55, 651-4. PMID: 17409565
- ↑ Lu J, Palmer BD, Kestell P, Browett P, Baguley BC, Muller G & Ching LM. (2003). Thalidomide metabolites in mice and patients with multiple myeloma. Clin. Cancer Res. , 9, 1680-8. PMID: 12738721
Reviews
Vargesson N. (2015). Thalidomide-induced teratogenesis: history and mechanisms. Birth Defects Res. C Embryo Today , 105, 140-56. PMID: 26043938 DOI.
Vargesson N. (2009). Thalidomide-induced limb defects: resolving a 50-year-old puzzle. Bioessays , 31, 1327-36. PMID: 19921660 DOI.
Gordon JN & Goggin PM. (2003). Thalidomide and its derivatives: emerging from the wilderness. Postgrad Med J , 79, 127-32. PMID: 12697909
Saunders EJ & Saunders JA. (1990). Drug therapy in pregnancy: the lessons of diethylstilbestrol, thalidomide, and bendectin. Health Care Women Int , 11, 423-32. PMID: 2228814 DOI.
Stephens TD. (1988). Proposed mechanisms of action in thalidomide embryopathy. Teratology , 38, 229-39. PMID: 3067416 DOI.
Newman CG. (1986). The thalidomide syndrome: risks of exposure and spectrum of malformations. Clin Perinatol , 13, 555-73. PMID: 3533365
Fletcher I. (1980). Review of the treatment of thalidomide children with limb defeciency in Great Britain. Clin. Orthop. Relat. Res. , , 18-25. PMID: 6991189
Articles
Benegbi M. (2007). 45 years later...where do we stand?. Can J Clin Pharmacol , 14, e37-9. PMID: 17213509 "The Thalidomide Victims Association of Canada (TVAC) was founded in 1988 and is the only organization in North America to work with and for Thalidomide victims. Our mission is to empower our members and to improve their quality of life through various programs and customized services. With the return of Thalidomide on the market, TVAC also took on the mandate of informing the public on the devastating effects of this medication and to promote awareness and caution when using any teratogenic products currently available".
SAUNDERS H, WRIGHT R & HODGKIN K. (1962). Thalidomide and congenital deformities. Br Med J , 2, 796. PMID: 14497528
LECK IM & MILLAR EL. (1962). Incidence of malformations since the introduction of thalidomide. Br Med J , 2, 16-20. PMID: 14463369
McBride WG. (1992). Prescription drugs in the first trimester and congenital malformations. Aust N Z J Obstet Gynaecol , 32, 386. PMID: 1290446
Books on Thalidomide
A selection of recent general public information books on Thalidomide, available from various internet commercial suppliers (search using the book title). Please note that this listing does not reflect an endorsement of the book or its content and is provided for educational purposes only.
- Thalidomide Kid (Paperback) by Kate, Rigby (Author)
- Thalidomide - A Medical Dictionary, Bibliography, and Annotated Research Guide to Internet References (Paperback) by ICON Health Publications (Author) Internet supplier link: Amazon
Search Pubmed
June 2010 "thalidomide teratogenicity" All (147) Review (57) Free Full Text (11)
Search Pubmed: thalidomide teratogenicity | Thalidomide | McBride WG |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- OMIM - CEREBLON | Thalidomide Susceptibility
- Medline Plus - Thalidomide
- USA FDA - Thalidomide
- Thalidomide Victims Association of Canada
- Thalidomide Isomerism and Optical Isomerism
- United Nations International Drug Control Programme
- Australian Drug Foundation (ADF)
- Centre for Education and Information on Drugs and Alcohol (CEIDA) (Australia)
- Child Health and Safety (Australia)
- NIDA (USA)- Consequences of Prenatal Drug Exposure
- Australian Medicines Handbook (no electronic version yet)
- Australian Congenital Anomalies Monitoring System (ACAMS)
- The Swedish Thalidomide Society
- Australia TGA - Advisory Committee on Medicines
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 19) Embryology Abnormal Development - Thalidomide. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Thalidomide
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G